Periodate Analysis.
After the separation, 1mL of NaIO4 0.02M (Fluka) was added to each fraction (60 drops/tube, 1.5 mL). Simultaneously, to a control tube with 1.5mL of distilled water, 1mL of periodate was added too. The tubes were kept in the oven at 30º C all night. After 12h, an aliquot of 100µL was taken from each tube and was diluted to 5mL (1/50 dilution).
Before the analysis, a solution of the complex (TPTZ)2Fe++ was prepared according to Pilch recommendations (54). The solution was diluted with AcOH/AcONa buffer pH 4.2 to an absorbance value of 0.8-0.9. at 593 nm
A 3.00 mL aliquot was taken to a standard spectrophotometric cuvette. This solution was titrated with the sample. Fixed volumes (usually 30µL or 40 µL)were added with a micropipette. After each addition the sample was homogeneized with a 1mL micropipette adjusted to 450µL until a stable reading was observed. A total of 6- 8 additions per sample were made.The fundamentals of the method are described next.
Lets consider succesive additions Vi mL of an oxidant to a Vo mL solution of a standard. The response of the apparatus (absorbance) diminish with each addition. We suppose an 1:1 stoichiometry and also suppose an irreversible reaction. The variation of absorbance will be:
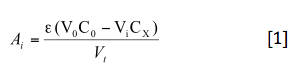
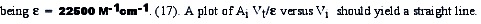
the concentration of periodate is obtained from the slope of the rect or from the equivalence volume.
The equivalence volume can be obtained by extrapolation to zero in the V axis.
In our case the reaction of oxidation is

So the final value is:
Cf = 1/2 f Cx [2]
being f the dilution factor (50 in our case)