SYNTHESIS OF ALLICIN
Allicin was synthesized by oxidation of diallyl disulfide with maganesium monoperoxy phthalate and tetrabutylammonium hydrogen sulfate as phase- transfer reagent
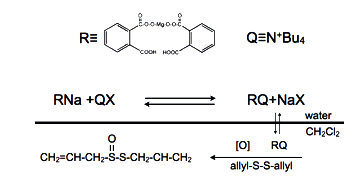
Scheme1. Oxidation of diallyl disulfide by phase-transfer process.
Diallyl disuldfide oxidation by magnesium monoperoxy phthalate yield allicin (1) as the oxidizr pass from aqueous to organic phase. But now we may question if quaternary ammonium salt behaves as a catalyst (2,3). The transfer of oxidant to organic phase should be accompanied of a lowering of the oxidation potential in the water phase. During the synthesis the redox potential in the aqueous phase was measured with a platinum electrode.Details of the apparatus are shown in Figure 1.

Figure 1. With this device both phases were simultaneously stirred a)reference electrode Ag/Ag/Cl ; b) platinum electrode c)glass rod; e) teflon stirrer;f) teflon base; g) magnetic bar; h) pH/volt meter; i) computer
To study the oxidant transfer to the organic phase three syntheses were made changing the concentration of initial quaternary ammonium salt (Q). The oxidizer MMPP (O) and diallyl disulfide were kept constant. The R values represent the ratio [Q]/[O]. During the synthesis the redox potential was monitored at regular time intervals.
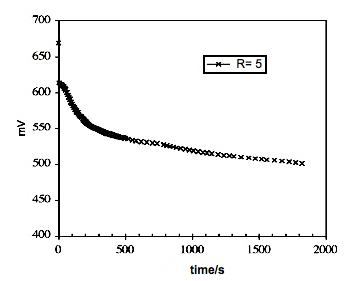
Figure2. Variation of redox potential during the synthesis of allicin, blank assay
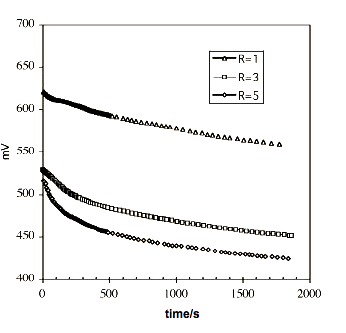
Figure3. Variation of redox potential for three R values
Figure 2 shows the variation of redox potential for a blank assay without disulfide in the organic phase. From t=0 to about 300s there is a steep descent in E originated by the transfer of the oxidant to the organic phase. The slight slope to the final time can be explained by a much lower rate of transfer, or decomposition of the peroxide in the water phase. In Figure 3 appears the variation of potential for three R values. The fall in the first five minutes is highest for R=5 followed by R=3 and R=1.These results suggest that the most oxidizer is transferred in the first five minutes and its amount depends on the initial ammonium sal concentration.
The amount of oxidant transferred was determined by potentiometric titration of MMPP in the aqueous phase before and after the synthesis. Sodium sulfite was used as titrant and a platinum electrode as indicator electrode.
The reaction is
2SO3= + R2(COOO)2Mg ----> 2S04= + 2RCOO- + Mg++
R being C6H4COO-
An example of titration curve is depicted in Figure4

Figure4. Potentiometric curves MMPP 20 mL titrated initial and after reaction for R=1 with numerical derivative
The analysis of MMPO allow us to know the amount of oxidizer transferred when the ratio [Q]/[O] was changed. These results are given in Table 1
Table1. Transfer of oxidant for three values of R
| Q, mmol | R | Ve,mL | T% |
| 1.03 | 1 | 8.46 | 63 |
| 3.09 | 3 | 6.75 | 70 |
| 5.15 | 5 | 5.15 | 77 |
| 5.15 | 5(blank) | 14.5 | 36 |
We can see that the fraction of MMPP tranferred depends on the initial ratio R. Moreover a five fold increase in R just produce a relative little increase of transfer to the organic phase. In the final assay, the initial amount of oxidant was doubled and the R1 value was let to 5. At the end of the synthesis an aliquot of the reaction was taken and analyzed by HPLC. In the third chromatogram of Figure 6 allyldisulfide is almost completely oxidized to allicin for the last set R=5 and R2 =1.5
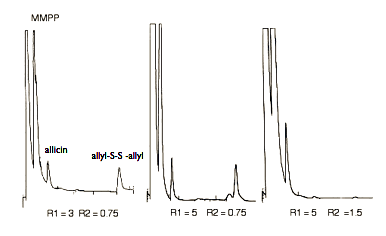
Figure6. Chromatograms of the reaction mixture for three different values of R 1 and R2 In the last chromatogram there is an almost complete conversion to allicin (notice the increase of the oxidizer peak MMPP)
From these results it is apparent that the synthesis of allicin does not follow the classical model of phase-transfer catalysis (4,5). Quaternary ammonium salt of the reduction product -phthalate- is lipophilic and very soluble in dichloromethane (see the first peak in the chromatogram). Moreover, allicin is unstable at room temperature so that the reaction must be done in ice bath. All these factors make difficult the transfer of ammonium salt from organic to aqueous phase after peroxide oxidation of disulfide.
In this kind of reactions the quaternary ammonium salt behaves like a carrier or surfactant rather than as a catalist, so that the chemist should adjust the initial concentrations of the reactants to obtain the optimal yield in the synthesis (6).
Literature cited
- Cruz Villalon G. Anal biochem.2001,290,376-378
- Gokel G.W.; Weber W. P. J. Chem Educ.1978,55,350-354
- Gokel G.W.; Weber W. P. J. Chem Educ.1978,55,429-433
- Starks C.J. J Am Chem Soc. 1971,93,195-199
- Starks C.J.;Owens R.M. J Am Chem Soc. 1973,95,3613-3617
- Cruz Villalon G. Anal Biochem.2002,304,274.







